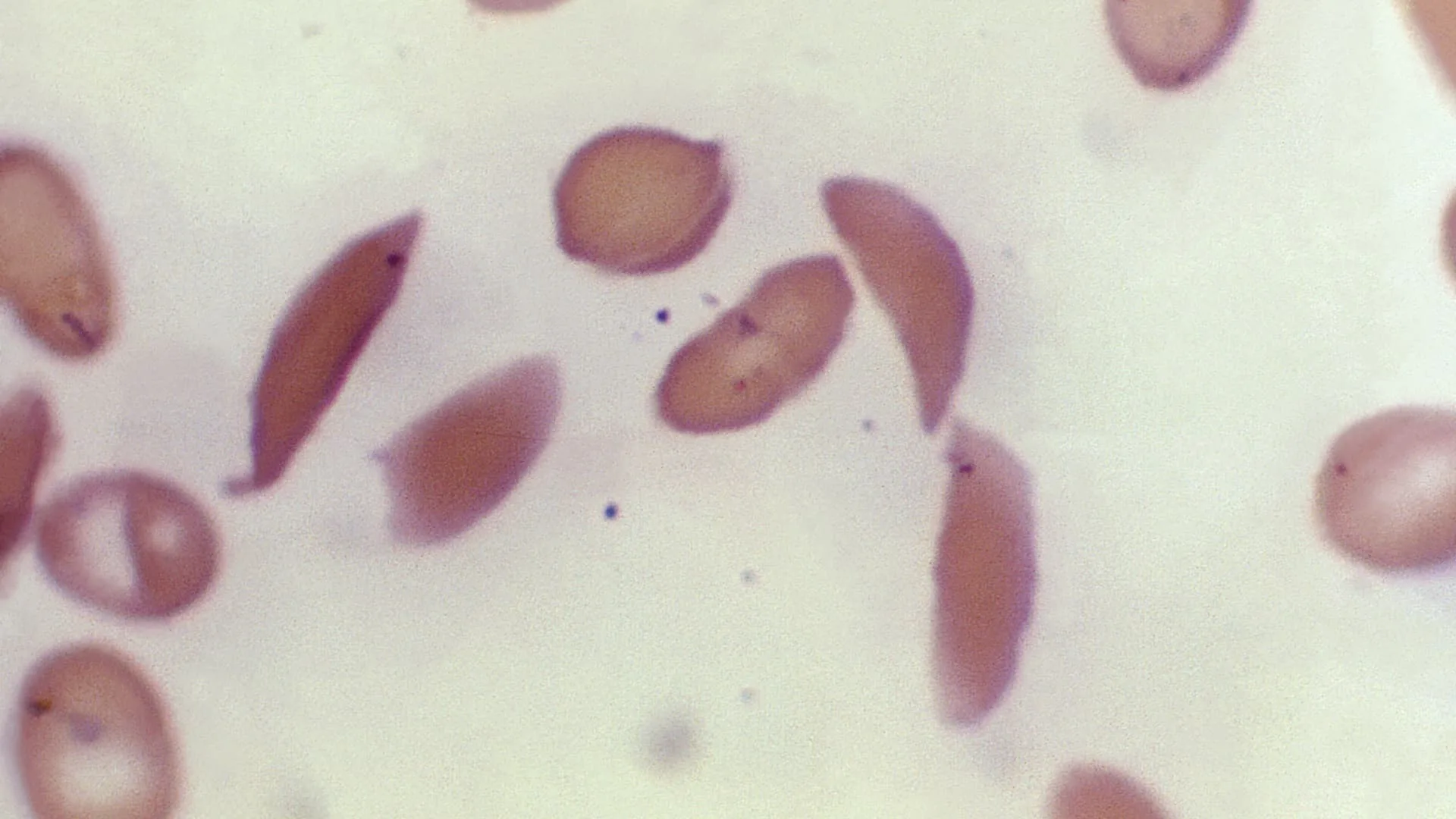
The U.S. Food and Drug Administration on Friday approved the country’s first gene-editing treatment, Casgevy, for use in patients with sickle cell disease. The approval comes about a decade after the discovery of CRISPR technology for editing human DNA, representing a significant scientific advancement. Yet reaching the tens of thousands of people who could benefit …
Continue reading “First CRISPR gene-editing remedy authorized in U.S.”