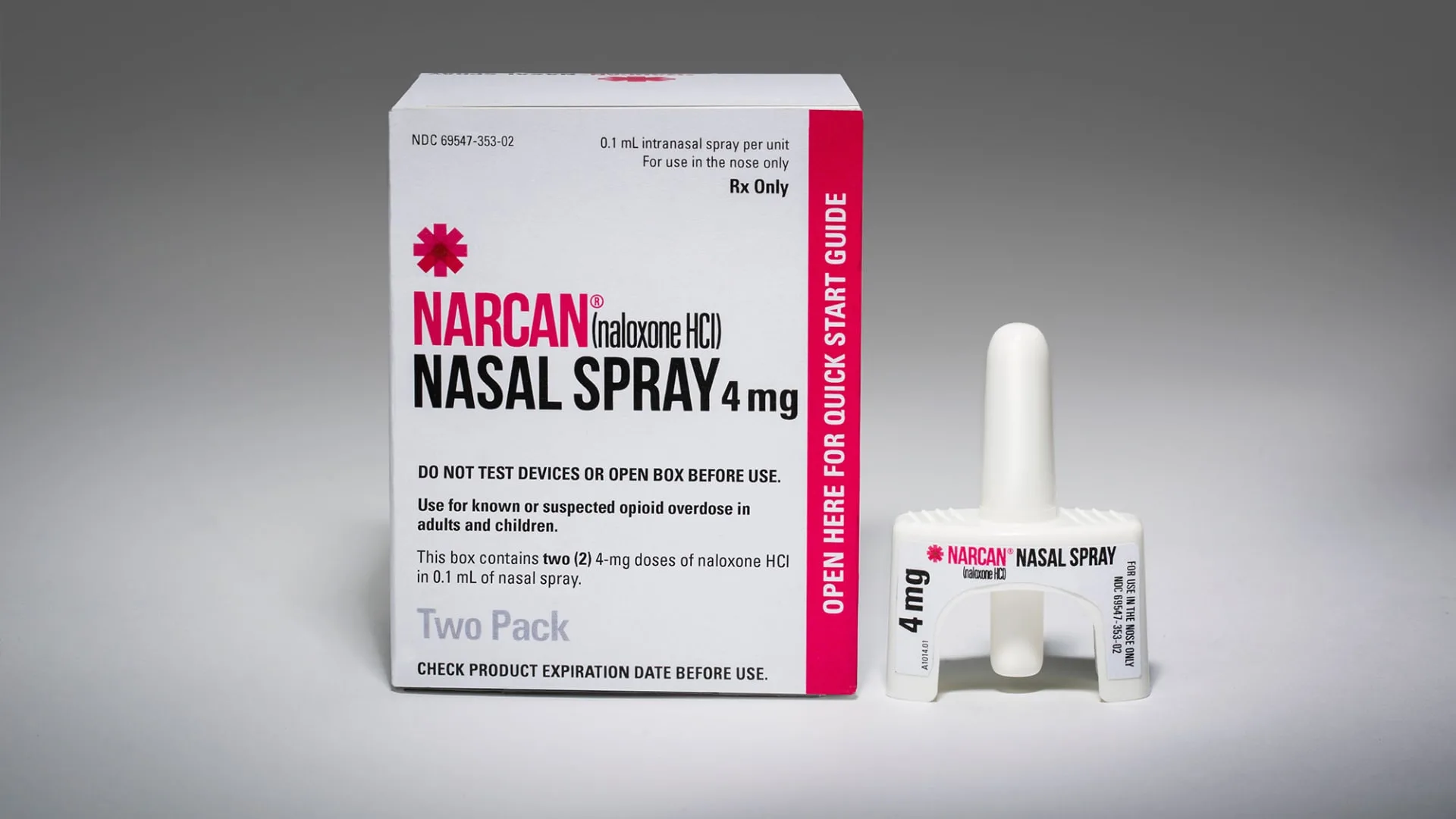
The independent advisors to the Food and Drug Administration on Wednesday unanimously recommended over-the-counter use of the nasal spray Narcan to reverse opioid overdoses, which would greatly expand access to the life-saving treatment.
Emerging biosolutions‘ Narcan is the top-selling treatment for opioid overdoses. The FDA is expected to make a decision by March 29 on whether people can buy the 4-milligram nasal spray without a prescription. The agency is under no obligation to accept the recommendation of its consultants, although it normally does.
“There’s no reason to keep this as a prescription, let’s get this out there and save some lives,” said Elizabeth Coykendall, a paramedic at PM Pediatrics in Raleigh, North Carolina, and a temporary voting member on the FDA committee.
Emergent BioSolutions said Narcan would be available for the over-the-counter market in late summer if the FDA approves it next month. The company has not yet revealed the cost of this.
“We’ve been working with key stakeholders like retailers and government officials on distribution plans,” said Matt Hartwig, a company spokesman.
Most states already have blanket prescriptions that allow pharmacies to dispense narcan, commonly known as naloxone, without requiring the patient to present a script. But FDA approval of Narcan for over-the-counter use would allow more people to more easily acquire the treatment in more places.
“When naloxone becomes a non-prescription product, it can be sold in many places that consumers have not previously had access to, including vending machines, convenience stores, supermarkets, and large department stores, just like other non-prescription products,” Jody Green, an official of the FDA’s nonprescription drug division, the advisory committee said Wednesday.
Since 1999, more than 564,000 people have died from opioids in three waves in the US — first from prescription opioids, then from heroin, and most recently from fentanyl, according to the Centers for Disease Control and Prevention. The number of opioid overdose deaths rose 17% during the pandemic, from about 69,000 in 2020 to nearly 81,000 in 2021.
The Trump administration first declared the opioid epidemic a public health emergency in 2017. The Biden administration has renewed the emergency declaration every 90 days since the president took office.
“Every day 187 people will die – that is absolutely tragic when we think not only of the individuals themselves but also of the families, the communities, the workplaces. This has profound human implications and it affects us all,” Manish Vyas, senior vice president of regulatory affairs at narcan-maker Emergent BioSolutions, told the committee.
Scott Hadland, chief of adolescent medicine at Massachusetts General Hospital, said the widespread infiltration of fentanyl into the nation’s drug supply has increased the risk of overdoses. Many people exposed to fentanyl take counterfeit pills that they thought were prescribed but actually contain the potent and often deadly opioid, Hadland said.
“And increasingly there are secondhand exposures that are also increasing,” Hadland, who attended the Emergent BioSolutions presentation, told the committee. “We are seeing rising overdose deaths in young children who encounter fentanyl in public settings, or fentanyl that may be elsewhere in the home.”
Hadland said he told parents to keep Narcan at their home in case of an emergency. He likened it to a fire extinguisher that families should have for safety but hopefully never have to use.
“Unfortunately, current access pathways are challenging for most young people, families and community members across the country,” Hadland said.
dr Bobby Mukkamala of the American Medical Association said Narcan should be as readily available as Tylenol, for treating headaches, or a decongestant for nasal congestion. Narcan should be as common in public places as AED machines used to treat people suffering heart attacks, Mukkamala said.
Jessica Hulsey, executive director of the Addiction Policy Forum, told the committee during a public comment section that if Narcan is sold over-the-counter, it must be offered at an affordable price of no more than $20 per dose. That’s because Narcan is packaged as unit doses, and multiple doses may be needed to reverse an overdose of potent fentanyl, Hulsey said.
Narcan displaces opioids, which bind to receptor sites in a person’s nervous system. By crowding out and blocking opioids, the nasal spray prevents fatal overdoses by reversing respiratory depression, said Gay Owens, head of global medical affairs at Emergent BioSolutions.
But Narcan needs to be administered as soon as an overdose is suspected, so it’s important to make sure the directions for using the nasal spray are simple, the FDA’s Green said. FDA advisors have worked to make the instructions on Narcan’s packaging as clear as possible so that anyone can easily use the device in a life-threatening emergency.
In a study sponsored by Emergent BioSolutions, more than 90% of 71 participants understood the instructions on the over-the-counter labels and used the Narcan device correctly during a simulated mannequin overdose emergency. Participants included people with different levels of reading and writing skills, as well as adults and young people.
However, some participants were confused by the five steps because they were spread across the side and back walls of the carton, said Millie Shah, senior pharmacist at the FDA’s division that oversees drug administration errors. This confusion, according to Shah, could result in delayed administration or failure to properly use the Narcan device when time is of the essence.
These instances occurred despite the fact that participants were given as much time as needed to familiarize themselves with Narcan’s instructions, which Shah says may not be the case in a real-life overdose emergency.
“As such, the data collected does not capture this highest-risk usage scenario,” Shah said.
The FDA has proposed that Emergent BioSolutions place all five instructions in sequence on the back of the carton and also include instructions in the device’s blister pack. The company presented a mockup at the advisory meeting, but the FDA said it hasn’t evaluated it yet.